Tripartite motif-containing protein 5
UniProt
Show FASTA Format
>sp|Q0PF16|TRIM5_MACMU Tripartite motif-containing protein 5 OS=Macaca mulatta OX=9544 GN=TRIM5 PE=1 SV=2
MASGILLNVKEEVTCPICLELLTEPLSLHCGHSFCQACITANHKKSMLYKEGERSCPVCR
ISYQPENIQPNRHVANIVEKLREVKLSPEEGQKVDHCARHGEKLLLFCQEDSKVICWLCE
RSQEHRGHHTFLMEEVAQEYHVKLQTALEMLRQKQQEAEKLEADIREEKASWKIQIDYDK
TNVSADFEQLREILDWEESNELQNLEKEEEDILKSLTKSETEMVQQTQYMRELISELEHR
LQGSMMDLLQGVDGIIKRIENMTLKKPKTFHKNQRRVFRAPDLKGMLDMFRELTDARRYW
VDVTLAPNNISHAVIAEDKRQVSSRNPQIMYQAPGTLFTFPSLTNFNYCTGVLGSQSITS
GKHYWEVDVSKKSAWILGVCAGFQSDAMYNIEQNENYQPKYGYWVIGLQEGVKYSVFQDG
SSHTPFAPFIVPLSVIICPDRVGVFVDYEACTVSFFNITNHGFLIYKFSQCSFSKPVFPY
LNPRKCTVPMTLCSPSS
Show Domain Info by CDD
Show Secondary Structure by PSIPRED
# PSIPRED HFORMAT (PSIPRED V3.2)
Conf: 973222101010115323112348743699934348874171113544799967776668
Pred: CCCHHHHCCCCCCCCCCCCCCCCCCCCCCCCCHHHHHHHCCCCCCCCCCCCCCCCCCCCC
AA: MASGILLNVKEEVTCPICLELLTEPLSLHCGHSFCQACITANHKKSMLYKEGERSCPVCR
10 20 30 40 50 60
Conf: 546899999983145899997723699877766511321030000131126815452022
Pred: CCCCCCCCCCCCHHHHHHHHHHHCCCCCCCCCCCCHHHHHCCCCCCCCCCCCEEEEECCC
AA: ISYQPENIQPNRHVANIVEKLREVKLSPEEGQKVDHCARHGEKLLLFCQEDSKVICWLCE
70 80 90 100 110 120
Conf: 354456973010789889989978999988777789997544567888989877444436
Pred: CCCCCCCCCEEEHHHHHHHHHHHHHHHHHHHHHHHHHHHHHHHHHHHHHHHHHHHHHHHH
AA: RSQEHRGHHTFLMEEVAQEYHVKLQTALEMLRQKQQEAEKLEADIREEKASWKIQIDYDK
130 140 150 160 170 180
Conf: 688999999998553898888643468999999688643567888679999999999998
Pred: HHHHHHHHHHHHHHHHHHHHHHHHHHHHHHHHHHHHHHHHHHHHHHHHHHHHHHHHHHHH
AA: TNVSADFEQLREILDWEESNELQNLEKEEEDILKSLTKSETEMVQQTQYMRELISELEHR
190 200 210 220 230 240
Conf: 256657862033588611223201599757701114544799078789845023550343
Pred: HCCCHHHHHCCHHHHHHCCCCCCCCCCCCCCCCCCCCCCCCCCHHHHHHHHHCCCCCEEE
AA: LQGSMMDLLQGVDGIIKRIENMTLKKPKTFHKNQRRVFRAPDLKGMLDMFRELTDARRYW
250 260 270 280 290 300
Conf: 578809999997122058881442598333479997545899898899985103378668
Pred: EEEEECCCCCCCCEEECCCCEEEECCCCCCCCCCCCCCCCCCCCCCCCCCCEECCCCCCC
AA: VDVTLAPNNISHAVIAEDKRQVSSRNPQIMYQAPGTLFTFPSLTNFNYCTGVLGSQSITS
310 320 330 340 350 360
Conf: 872389962898602752026874584456686301259998589997379837660359
Pred: CCCEEEEECCCCCCEEEECCCCCCCCCCCCCCCCCCCCCCCCEEEEEECCCCEEEECCCC
AA: GKHYWEVDVSKKSAWILGVCAGFQSDAMYNIEQNENYQPKYGYWVIGLQEGVKYSVFQDG
370 380 390 400 410 420
Conf: 999999972123237469992489701678811342046898116742899999976346
Pred: CCCCCCCCCCCCCCCCCCCCEEEEEEECCCCCEEEEECCCCCCEEEEECCCCCCCCCCCC
AA: SSHTPFAPFIVPLSVIICPDRVGVFVDYEACTVSFFNITNHGFLIYKFSQCSFSKPVFPY
430 440 450 460 470 480
Conf: 36999999926419999
Pred: CCCCCCCCCCEECCCCC
AA: LNPRKCTVPMTLCSPSS
490
Show Conservation Score by AL2CO
1 M 3.357
2 A 1.287
3 S 0.209
4 G -0.445
5 I -0.631
6 L -0.280
7 L -0.497
8 N -0.400
9 V 0.132
10 K -0.540
11 E 0.177
12 E 0.967
13 V 0.371
14 T 0.135
15 C 3.357
16 P 1.201
17 I 1.534
18 C 3.357
19 L 0.621
20 E 0.272
21 L -0.076
22 L 0.680
23 T -0.160
24 E 0.905
25 P 2.148
26 L 0.817
27 S 0.371
28 L 0.979
29 H -0.445
30 C 3.057
31 G 1.694
32 H 3.357
33 S 0.661
34 F 1.401
35 C 3.067
36 Q 0.056
37 A -0.300
38 C 3.357
39 I 1.785
40 T -0.460
41 A -0.744
42 N -0.318
43 H -0.311
44 K -0.106
45 K -0.593
46 S -0.063
47 M -0.855
48 L -0.875
49 Y -0.853
50 K -0.566
51 E -0.306
52 G -0.704
53 E -0.774
54 R -0.671
55 S -0.443
56 C 3.357
57 P 2.372
58 V 0.073
59 C 2.829
60 R 0.824
61 I -0.786
62 S -0.652
63 Y -0.131
64 Q -0.524
65 P -0.668
66 E -0.475
67 N -0.278
68 I 0.280
69 Q 0.142
70 P -0.287
71 N 1.660
72 R -0.099
73 H -0.078
74 V 1.264
75 A 0.378
76 N 0.268
77 I 1.002
78 V 0.548
79 E 0.225
80 K -0.383
81 L 0.397
82 R -0.167
83 E -0.704
84 V -0.513
85 K -0.728
86 L -0.460
87 S -0.882
88 P -0.460
89 E -0.661
90 E -0.846
91 G -0.630
92 Q -0.476
93 K -0.644
94 V -0.590
95 D -0.922
96 H -0.656
97 C 2.832
98 A -0.740
99 R -0.761
100 H 3.085
101 G 0.001
102 E 1.333
103 K -0.656
104 L 1.094
105 L -0.329
106 L 0.316
107 F 2.237
108 C 3.357
109 Q -0.201
110 E 0.137
111 D 1.979
112 S -0.121
113 K -0.357
114 V 0.236
115 I 0.792
116 C 2.829
117 W -0.629
118 L -0.070
119 C 3.357
120 E -0.459
121 R -0.691
122 S 0.620
123 Q -0.382
124 E 0.027
125 H 2.679
126 R -0.144
127 G -0.498
128 H 3.074
129 H -0.730
130 T 0.112
131 F -0.463
132 L 0.697
133 M 0.312
134 E 0.263
135 E 1.095
136 V 1.045
137 A 0.484
138 Q -0.675
139 E -0.613
140 Y 0.385
141 H 1.041
142 V -0.404
143 K -0.048
144 L 0.861
145 Q -0.095
146 T -0.576
147 A -0.648
148 L 0.345
149 E -0.491
150 M -0.908
151 L 0.823
152 R -0.201
153 Q -0.246
154 K -0.289
155 Q -0.427
156 Q -0.434
157 E -0.630
158 A -0.138
159 E -0.593
160 K -0.547
161 L -0.299
162 E -0.454
163 A -0.693
164 D -0.843
165 I -0.612
166 R -0.478
167 E -0.377
168 E -0.056
169 K -0.639
170 A -0.594
171 S -0.701
172 W 0.116
173 K 0.264
174 I -0.785
175 Q -0.421
176 I 0.073
177 D -0.148
178 Y -0.956
179 D -0.146
180 K -0.162
181 T -0.380
182 N -0.707
183 V 0.666
184 S -0.697
185 A -0.847
186 D -0.081
187 F 0.766
188 E -0.434
189 Q -0.313
190 L -0.019
191 R -0.371
192 E -0.647
193 I -0.246
194 L 1.597
195 D -0.664
196 W -0.725
197 E -0.037
198 E 0.969
199 S -0.549
200 N -0.626
201 E -0.422
202 L 0.315
203 Q -0.481
204 N -0.386
205 L 1.513
206 E -0.398
207 K -0.271
208 E -0.008
209 E 0.206
210 E -0.314
211 D -0.698
212 I -0.570
213 L -0.355
214 K -0.807
215 S -0.884
216 L 0.517
217 T -0.629
218 K -0.317
219 S -0.306
220 E -0.621
221 T -0.652
222 E -0.713
223 M 0.346
224 V -0.738
225 Q -0.544
226 Q 0.001
227 T -0.467
228 Q -0.678
229 Y -0.764
230 M 0.594
231 R -0.547
232 E -0.470
233 L -0.177
234 I 0.950
235 S -0.434
236 E 0.050
237 L 0.396
238 E 0.437
239 H -0.662
240 R -0.270
241 L -0.535
242 Q -0.494
243 G -0.759
244 S -0.031
245 M -0.471
246 M -0.674
247 D -0.655
248 L 0.310
249 L 1.010
250 Q -0.136
251 G -0.110
252 V -0.431
253 D -0.545
254 G -0.814
255 I -0.525
256 I -0.597
257 K -0.834
258 R -0.239
259 I -0.714
260 E -0.759
261 N -0.984
262 M -0.882
263 T -0.760
264 L -0.749
265 K -0.742
266 K -0.787
267 P -0.711
268 K -0.886
269 T -0.793
270 F -0.837
271 H -0.843
272 K -0.878
273 N -0.827
274 Q -0.390
275 R -0.438
276 R -0.856
277 V -1.021
278 F -0.841
279 R -0.992
280 A -0.752
281 P -0.692
282 D -0.904
283 L -0.869
284 K -0.932
285 G -0.964
286 M -0.690
287 L -0.897
288 D -0.813
289 M -1.077
290 F -0.919
291 R -0.944
292 E -0.820
293 L -0.884
294 T -0.914
295 D -0.977
296 A -0.849
297 R -0.901
298 R -1.017
299 Y -0.887
300 W -0.852
301 V -0.449
302 D -0.758
303 V -0.183
304 T -0.541
305 L 0.127
306 A -0.377
307 P -0.114
308 N -0.767
309 N -0.305
310 I -0.276
311 S -0.632
312 H -0.616
313 A -0.591
314 V -0.512
315 I -0.211
316 A -0.041
317 E -0.337
318 D 0.250
319 K -0.682
320 R -0.071
321 Q -0.551
322 V 0.046
323 S -0.507
324 S -0.792
325 R -0.732
326 N -0.810
327 P -0.849
328 Q -0.748
329 I -0.856
330 M -1.033
331 Y -0.582
332 Q -0.775
333 A -1.074
334 P -0.774
335 G -1.074
336 T -1.000 *
337 L -1.000 *
338 F -1.000 *
339 T -1.000 *
340 F -0.958
341 P -1.034
342 S -0.590
343 L -0.782
344 T -0.900
345 N -0.294
346 F -0.329
347 N -0.807
348 Y -1.011
349 C -0.821
350 T -0.642
351 G -0.565
352 V 0.935
353 L 1.118
354 G 1.810
355 S -0.405
356 Q 0.091
357 S -0.864
358 I 1.202
359 T -0.623
360 S 0.566
361 G 2.185
362 K 1.289
363 H 0.067
364 Y 1.472
365 W 3.357
366 E 1.426
367 V 1.732
368 D -0.023
369 V 2.116
370 S -0.258
371 K -0.607
372 K 0.381
373 S -0.837
374 A -0.678
375 W 2.830
376 I -0.351
377 L 1.341
378 G 3.357
379 V 1.331
380 C 0.194
381 A -0.668
382 G -0.495
383 F -0.861
384 Q -0.955
385 S -1.000 *
386 D -0.835
387 A -0.886
388 M -0.875
389 Y -0.953
390 N -0.817
391 I -0.695
392 E -0.633
393 Q -0.614
394 N -1.100
395 E -0.801
396 N -0.962
397 Y -0.477
398 Q -0.622
399 P 0.100
400 K -0.598
401 Y -0.794
402 G 1.394
403 Y 0.501
404 W 2.030
405 V -0.055
406 I 0.959
407 G -0.219
408 L 0.553
409 Q -0.855
410 E -0.615
411 G -0.588
412 V -0.835
413 K -0.472
414 Y 0.919
415 S -0.751
416 V 0.576
417 F -0.179
418 Q -0.097
419 D -0.648
420 G -0.231
421 S -0.618
422 S -0.769
423 H -0.649
424 T -1.000 *
425 P -1.000 *
426 F -1.000 *
427 A -0.714
428 P -1.000 *
429 F -0.839
430 I -0.816
431 V 0.099
432 P -0.240
433 L 0.878
434 S -0.505
435 V 0.192
436 I -0.893
437 I -0.755
438 C -0.619
439 P 1.360
440 D -0.595
441 R -0.069
442 V 1.470
443 G 1.983
444 V 2.127
445 F 0.719
446 V 2.103
447 D 1.843
448 Y 0.622
449 E 0.584
450 A -0.233
451 C 0.284
452 T -0.305
453 V 1.601
454 S 1.183
455 F 2.890
456 F 1.091
457 N 1.667
458 I 0.485
459 T 0.251
460 N 0.313
461 H -0.672
462 G 0.087
463 F -0.250
464 L 0.702
465 I 1.469
466 Y 1.446
467 K 0.026
468 F 1.643
469 S -0.351
470 Q -0.616
471 C -0.629
472 S -0.751
473 F 1.813
474 S -0.515
475 K -0.446
476 P -0.706
477 V 0.536
478 F -0.070
479 P 2.238
480 Y 1.022
481 L 2.050
482 N -0.075
483 P 0.196
484 R -0.187
485 K -0.951
486 C -0.276
487 T -0.704
488 V -0.781
489 P 0.877
490 M 1.127
491 T -0.076
492 L 1.394
493 C 1.028
494 S -0.340
495 P 0.735
496 S 0.029
497 S -1.000 *
* gap fraction no less than 0.50; conservation set to M-S
M: mean; S: standard deviation
al2co - The parameters are:
Input alignment file - 244.paln
Output conservation - STDOUT
Weighting scheme - independent-count based
Conservation calculation method - entropy-based
Window size - 1
Conservation normalized to zero mean and unity variance
Gap fraction to suppress calculation - 0.50
10 20 30 40 50 60
MASGILLNVK EEVTCPICLE LLTEPLSLHC GHSFCQACIT ANHKKSMLYK EGERSCPVCR
70 80 90 100 110 120
ISYQPENIQP NRHVANIVEK LREVKLSPEE GQKVDHCARH GEKLLLFCQE DSKVICWLCE
130 140 150 160 170 180
RSQEHRGHHT FLMEEVAQEY HVKLQTALEM LRQKQQEAEK LEADIREEKA SWKIQIDYDK
190 200 210 220 230 240
TNVSADFEQL REILDWEESN ELQNLEKEEE DILKSLTKSE TEMVQQTQYM RELISELEHR
250 260 270 280 290 300
LQGSMMDLLQ GVDGIIKRIE NMTLKKPKTF HKNQRRVFRA PDLKGMLDMF RELTDARRYW
310 320 330 340 350 360
VDVTLAPNNI SHAVIAEDKR QVSSRNPQIM YQAPGTLFTF PSLTNFNYCT GVLGSQSITS
370 380 390 400 410 420
GKHYWEVDVS KKSAWILGVC AGFQSDAMYN IEQNENYQPK YGYWVIGLQE GVKYSVFQDG
430 440 450 460 470 480
SSHTPFAPFI VPLSVIICPD RVGVFVDYEA CTVSFFNITN HGFLIYKFSQ CSFSKPVFPY
490
LNPRKCTVPM TLCSPSS
2LM3 (NMR)
4B3N (X-Ray,3.3 Å resolution)
4TKP (X-Ray,2.08 Å resolution)
5EIU (X-Ray,1.91 Å resolution)
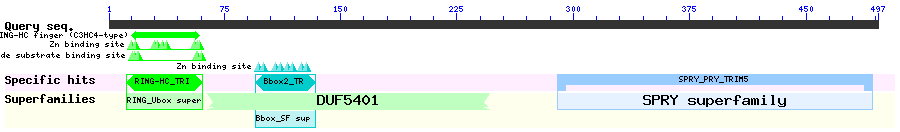
The splicing variant alpha of TRIM5 is composed of four distinct domains: RING, B-box 2, coiled-coil and B30.2(SPRY) domains. TRIM5alpha is a retrovirus restriction factor, which mediates species-specific, early block to retrovirus infection. Multiple studies illustrated LMB sensitivity of the protein. Diaz-Griffero et al. performed N-terminal deletion studies and showed that deletion of 1-93 caused nuclear accumulation while deletion of 1-60 didn't, suggesting that 60-93 is sufficient to direct nuclear exit. There is no clear NES consensus match based on leucines, however there are valines and leucines throughout the region. This region appears to take on loop-alpha helix structure until residues 81 and residues beyond 81 is unstructured and is likely the linker region that connects to the following B-box ZnF.
[1]. "Role of SUMO-1 and SUMO interacting motifs in rhesus TRIM5alpha-mediated
restriction"
Lukic Z, Goff SP, Campbell EM, Arriagada G. (2013)
Retrovirology,
10(1):
PubMed[2]. "Contribution of SUMO-interacting motifs and SUMOylation to the antiretroviral properties of TRIM5alpha"
Brandariz-Nuñez A, Roa A, Valle-Casuso JC, Biris N, Ivanov D, Diaz-Griffero F. (2013)
Virology,
435(2):463-71
PubMed[3]. "The Conserved Sumoylation Consensus Site in TRIM5α Modulates Its Immune Activation Functions"
Nepveu-Traversy MÉ, Berthoux L. (2014)
Virus Res,
184:30-8
PubMed[4]. "Trafficking of some old world primate TRIM5α proteins through the nucleus"
Diaz-Griffero F, Gallo DE, Hope TJ, Sodroski J. (2011)
Retrovirology,
8:38
PubMed
Accurate identification of NESs is difficult because many sequences in the genome match the NES consensus.
Therefore, some published NESs may be mistakenly identified. Please help us improve the accuracy of NESdb
by providing either a positive or negative flag for the NES in this entry. Supporting comments are required to process the flag.
